HeaRTNovation
TAYÔ Project – Robotic Rehabilitation for the Trunk and Lower Extremity: STAGE 2 – Safety Tests, Performance Evaluation, and Phase 1 of the Clinical Trial
Technology Generator
De La Salle University
Project leader: Dr. Armyn C. Sy
The Problem
Stroke and injury are two of the leading causes of long-term disability worldwide. A solution to treat these stroke or injury sufferers is rehabilitation through physical therapy.
Due to limited rehabilitation devices and labor-intensive conventional therapies, research and development on lower limb rehabilitation robots has been continuously growing. Through the years, it was concluded that usage of robots as therapy aid devices give efficient rehabilitation similar to manual, conventional practices. However, translating these researches into commercially-viable products and reaching the end-users have been a problem. Moreover, although proven efficient, the role of robotic rehabilitation devices or exoskeletons remain to be supplementary rather than necessary.
The Solution
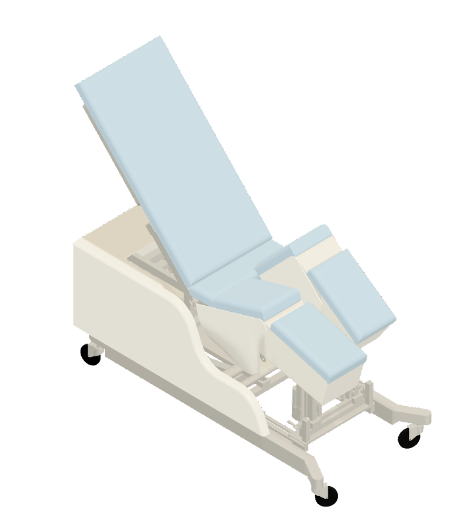 The Institute of Biomedical Engineering and Health Technologies (IBEHT), under De La Salle University, and in collaboration with the De La Salle Medical and Health Sciences Institute, developed and designed TAYO, a robotic rehabilitation for the trunk and lower extremity. TAYO would feature a multifunctional system that can move from supine to sitting or sitting to supine positions and has surface electromyography (sEMG) to quantify patients’ ability and progress. The device allows range of motion (ROM) exercises to be performed while the patient is in supine or sitting positions and through passive, active-assist, or active-resist (strengthening) training modes. The ROMs include hip flexion-extension, hip abduction-adduction, knee flexion-extension, trunk flexion, and trunk lateral flexion. Furthermore, games using Kinect v2.0 sensors were developed to help motivate patients to perform such gait training exercises.
The Institute of Biomedical Engineering and Health Technologies (IBEHT), under De La Salle University, and in collaboration with the De La Salle Medical and Health Sciences Institute, developed and designed TAYO, a robotic rehabilitation for the trunk and lower extremity. TAYO would feature a multifunctional system that can move from supine to sitting or sitting to supine positions and has surface electromyography (sEMG) to quantify patients’ ability and progress. The device allows range of motion (ROM) exercises to be performed while the patient is in supine or sitting positions and through passive, active-assist, or active-resist (strengthening) training modes. The ROMs include hip flexion-extension, hip abduction-adduction, knee flexion-extension, trunk flexion, and trunk lateral flexion. Furthermore, games using Kinect v2.0 sensors were developed to help motivate patients to perform such gait training exercises.
Project Team
Under the IBEHT, the TAYO project partners with De La Salle Medical and Health Sciences Institute (DLSHSI) to develop a cost-effective, multi-functional robotic device for early trunk and lower limb rehabilitation. With the collaboration of these two institutions, the project hopes to foster the development of rehabilitation devices as a center of medical device research for engineers and researchers.
Product Development Stage
The device is currently at Technology Readiness Level (TRL) 5. The system has successfully completed all safety testing, including mechanical testing, electrical testing, and Electro-Magnetic Compatibility (EMC) testing, to ensure conformity with local and international safety and compliance standards. A clinical trial with healthy human participants is currently ongoing, having received clearance from the Ethics Review Board (ERB). The research team is also seeking potential R&D partners in the areas of robotics rehabilitation, assistive technologies, and biomechanics
Contact Information
Dr. Nilo T. Bugtai
Director, Institute of Biomedical Engineering and Health Technologies
nilo.bugtai@dlsu.edu.ph




